National Depression Glass Association
Preserving America's Glass Manufacturing Heritage
Chemistry of Glass
by Bettye Waher
Glass Review - January 1979
For centuries glass has fascinated, intrigued, and lured men into desiring it. So much so that it is one of the earliest man-made substances, probably the first. Its charm has caused it to be produced in a great variety of forms and kinds of decoration, much of it of great beauty. In fact, the earliest glass was made for an esthetic reason rather than practical; for from Egypt came small glass-covered stones (beads) dating from about 2500 B.C. The Egyptians also used glass as an inlay to adorn furniture and other articles.
Glass as an artistic medium has flourished only in times of prosperity, and the course of glass has followed the history of each succeeding civilization: from the shores of the Mediterranean, where it had its beginning; carried by the Phoenicians to Europe; and from there to the New World. Glass was apparently the first industry to be transplanted into the New World for by 1535 glass was being made in Mexico. Then in 1608, The London Company of Virginia set up a glass house in Jamestown. Thus, as man moved and established new civilizations, he carried this love of glass with him.
The charm of glass is not only in its great beauty but also in the wide range of things that can be done to it and with it. It has been molded, blown, pressed, and decorated in a myriad of ways: colored, enameled, cut, engraved, layered, etched, gilded, silvered, stained, applied to itself and as a glaze onto other materials. So by presenting this challenge of seemingly unlimited possibilities, it has furthered its charm.
But its utilitarian value is also part of its charm. It intrigues with the question: how could a material so beautiful also be so serviceable? Glass is a container, windowpane, mirror, lens, scientific apparatus, fabric, insulator, tableware, construction brick, and material to replace steel in car bodies.
Thus glass has charmed by performing many roles: the dramatic, as a brilliant cut piece; the comedian, as a "whimsey"; and as the helpful servant. It has appeared in literature and has even been a child's toy. Artful, beautiful and dutiful, this is the charm of glass.
But before glass could be all of these things and do all these things for man, man had to make it. Gold, silver, and diamonds came from nature in an almost ready to use state, but glass had to be produced, for glass as we know it does not exist in nature. The nearest natural substances are obsidian, a "glass-like" volcanic quartz, and rock crystal, a transparent and colorless quartz. Which was man attempting to reproduce? It is impossible to answer that question, but he actually came nearer to reproducing obsidian. Both glass and obsidian are non-crystalline while rock crystal has a crystalline structure.
When did man first discover that he could turn sand into glass? When did he learn that in much the same way that volcanic heat produced obsidian from silica, he too could make glass? Another question without a precise answer, but it is believed that sometime between 4000 and 3000 B.C. glass was being made along the shores of the Mediterranean -- probably in Egypt.
Although the above questions remain unanswered, how glass is made and the chemistry of this marvelous product is known. Chemistry is the study of the composition and the changes, physical and chemical, in matter. When this definition is applied to glass, the following facts are found.
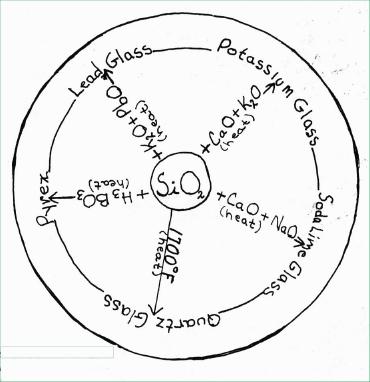 Composition: the principal
element in glass is silicon (Si) but silicon does not occur free in
nature, it occurs only as a compound. Silicon compounds make up 87% of
the earth's surface, the most common being silica (silicon dioxide,
Si02). Silica is found extensively in nature as quartz and
sand. Sometimes it is colored by the presence of other substances, as
in smoky quartz, rose quartz, amethyst, onyx, jasper. Opal and flint
are hydrated (contain water) forms. Silica melts at 1700°F to form
a viscous liquid that upon cooling forms quartz glass. This type of
glass is used in making many forms of laboratory glassware because it
will withstand temperatures up to 1500°F before softening.
Composition: the principal
element in glass is silicon (Si) but silicon does not occur free in
nature, it occurs only as a compound. Silicon compounds make up 87% of
the earth's surface, the most common being silica (silicon dioxide,
Si02). Silica is found extensively in nature as quartz and
sand. Sometimes it is colored by the presence of other substances, as
in smoky quartz, rose quartz, amethyst, onyx, jasper. Opal and flint
are hydrated (contain water) forms. Silica melts at 1700°F to form
a viscous liquid that upon cooling forms quartz glass. This type of
glass is used in making many forms of laboratory glassware because it
will withstand temperatures up to 1500°F before softening.
All glass begins by fusing silica and then the composition is varied depending on the use for which the glass is intended. The glass used in making bottles and window panes is "Soda glass" containing silica, sodium oxide (NaO), and calcium oxide (CaO). The CaO is necessary to prevent water solubility. Soda glass is sometimes called "Soft Glass" because it will soften enough at relatively low temperatures. Potassium glass or "Hard Glass" softens at higher temperatures and differs in composition from soda glass in that potassium oxide (K20) is used in place of NaO. "Flint glass," more properly called lead glass, is a potassium glass in which the CaO has been replaced by lead oxide (PbO). It is a heavy, very brilliant, durable glass that more closely resembles rock crystal. It is used in making the finest quality glassware and for lenses in optical equipment. A glass known by the trade names "Pyrex" and "Fire king" is made by fusing silica with boric ad (H3B03). It has a low coefficient of expansion so it is suitable for laboratory glassware and ovenware. A special process developed in Great Britain called "titanizing" (treating the glass surface with a titanium (Ti) compound) results in the increase in the working life of returnable bottles, due to reduction of friction and increase in the resistance to tiny cracks in the treated glass.
 By the addition
of different substances to the molten charge, called the metal, colored
glass can be obtained. Ferrous (Iron, Fe) compounds give green; cobalt
(Co) gives blue; manganese (Mn) produces violet to purple; carbon (C)
gives dark yellow to brown; selenium (Se) and gold (Au) yield the reds;
manganese and iron used together give black; and stannic (tin, Sn)
oxide gives an opaque white called "milk glass."
By the addition
of different substances to the molten charge, called the metal, colored
glass can be obtained. Ferrous (Iron, Fe) compounds give green; cobalt
(Co) gives blue; manganese (Mn) produces violet to purple; carbon (C)
gives dark yellow to brown; selenium (Se) and gold (Au) yield the reds;
manganese and iron used together give black; and stannic (tin, Sn)
oxide gives an opaque white called "milk glass."
Physical properties: the general usefulness and beauty of glass is due to an unique combination of physical and chemical properties possessed by no other substance. Glass is an amorphous (non-crystalline) compound. A homogeneous beam of x-rays can show the internal structure of a crystal. By measurement of the lines obtained on a photographic film, conclusions may be drawn both as to the structure and size of the crystals, But truly amorphous substances such as glass give none of the characteristic lines of crystals. In crystalline substances the atoms are linked in an orderly arrangement while an amphorous substance has an at random arrangement. Any material that cools from a liquid state to a rigid condition without crystallization is properly called a glass, but it must be cooled gradually (annealed). if cooled too quickly, it reaches ordinary temperature in an unstable condition and is extremely hard and brittle as compared to glass that has been annealed properly. The giant 200 inch curved mirror reflector in the Mount Palomar telescope was annealed for two years. Improper annealing causes imperfect fusion of the components and minute fissures can be seen in the glass, this is one type of so-called "sick glass." The annealing process is necessary because glass has no definite melting point but gradually softens, and likewise it has no definite freezing point but gradually gels, the outer surface setting up first. It has a smooth cooling curve without the sharp break which indicates the freezing point of a liquid that crystallizes at a definite temperature.
Other physical properties: glass is not reflective, light travels through clear glass, but it can be made reflective by curving or putting something reflective behind it such as silvering the back of a mirror. Glass is refractive (will bend light rays), the extent of refraction depends upon the different wavelengths and upon the different types of glass. The refractive index for fused silica is 1.459, soda-lime glass is 1.512 and lead glass is 1.560. The high refractive ability of lead glass explains its brilliance and its use in microscopes and telescopes. Glass expands when heated, shrinks when cooled, and is a poor conductor of heat and electricity.
Chemical properties: glass is relatively inactive, but three of its chemical reactions are of particular importance. Pure silica is insoluble in water but it has a poor resistance to alkalies. Thus soda-lime glass contains the agent of its own destruction for both soda and lime are alkalies. When soda-lime glass has been in use for a time, moisture in the air begins to leach sodium ions from the surface, these form sodium hydroxide (NaOH) an alkali that attacks the silica in the glass. The microscopic fissures this creates give a whitish look to the glass, another type of "sick glass." The reaction of glass with carbon dioxide (CO2) in the air can also cause deterioration of glass surfaces when moisture is present. The third reaction of importance is that of glass with hydrofluoric acid (HF) which is used in the etching, polishing and signing of glass.
In etching, the glass article is first covered with an acid-resistant film, then the design is scribed through the coating and then the article is dipped in hydrofluoric acid which "eats" the design out of the surface of the glass. The reaction that takes place is that the HF dissolves the silica in the glass evolving it as silicon tetrafluoride (SiF4) gas. The equation for this reaction is: Na2Si3O7 -- CaSi307(soda-lime glass) + 28HF --> 2NaF + CaF2 + 6SiF4 + 14H20. Basically this is the same reaction that takes place when HF is used for polishing glass. The glass article is dipped in a HF bath to "cure" the blemishes of cutting and to restore the brilliance. After being cut, the surface of the glass takes on a smoky or whitish look and this has to be polished away. Up until about the turn of the century, all polishing was done by wheels along with pumice (hand polishing) but the acid bath was found to be quicker and cheaper. However, it is very dangerous -- the worker has to wear protective clothing and a face mask. Great care must be taken in the time the article is in the bath and also the strength of the bath must be accurate or too much of the silica will be dissolved and the glass will be pitted. (Many master cutters felt that acid polished articles never have the brilliancy of the hand polished.)
This exploration of the charm and chemistry of glass reveals that glass is both a fine art and an exact science. Many glass pieces are to be found in museums and possibly the museum building is constructed of glass bricks and insulated with fiberglass. Therein lies its charm, this range from museum piece to building construction. But before it could be either, it had to be produced and its production was dependent upon its chemistry.
Bibliography:
Baskerville, Charles and Curlman, Louis - A Course in Qualitative
Chemical Analysis. MacMillan Co.; New York, NY; 1925.
Boggess, Bill and Louise - American Brilliant Cut Glass. Crown
Publishing, Inc.; New York, NY; 1977.
Brinkley, Stuart R. - Principles of General Chemistry,
MacMillan Co.; New York, NY; 1941
Colliers Encyclopedia, Vol. II - Crowell-Collier Educational
Corp., U.S.A.; 1973
Compton's Encyclopedia, Vol.9 - F, E. Compton Co.; Chicago,
IL.; 1972
Oiiver, Elizabeth - American Antique Glass. Golden Press; New
York, NY; 1977
The New Encyclopaedia Britannica, Vol.8 - Encyclopaedia
Brittanica, Inc.; London 1975
Editors note: About the author: Bettye W. Waher has a B. S. degree from Western Kentucky University, worked ten years as a chemist and taught science and math for fourteen years. Now retired, Bettye is operating her business "The Hawkes Hunter" from her home.